![[headshot]](https://cdn.prod.website-files.com/68e9289a324a09160f2cdcc4/69826868dfda88e10069cb44_Betsy%20(2).png)
.png)
Subscribe our newsletter to receive the latest blogs, case studies and publications.
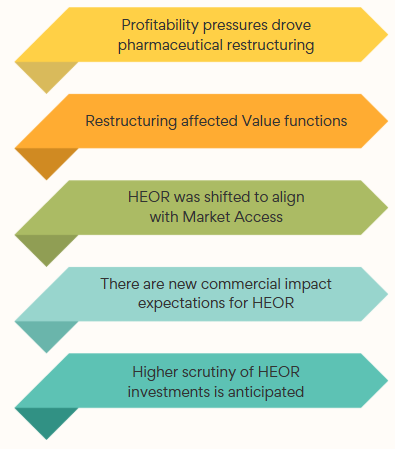
SubscribeExhibit 1: Restructuring Key Findings
Pharmaceutical manufacturers are facing significant business headwinds. In 2024, we observed major reorganizations. To better understand the rationale and impact of these changes for Health Economics and Outcomes Research (HEOR) and Market Access teams, we interviewed subject matter experts (SMEs) in these “Value” functions.
Participants shared perspectives on the factors driving restructuring. They reported changes in organizational design, new commercial impact expectations, and higher scrutiny (Exhibit 1).
Value functions, particularly HEOR, must influence evidence development and market shaping investments throughout the product lifecycle to deliver expected commercial impact. While securing budgets in large organizations challenges many departments, inadequate resourcing for product value strategies poses a material risk. This can compromise both current launches and future innovation.
To support leaders advocating for HEOR resources in new organizations, we recommend the Impact Communication Framework and Value Dashboard Tool.
The objectives of this initiative were to:
Our method (Exhibit 2) consisted of recruiting subject matter SMEs in Value functions who were familiar with recent pharmaceutical organization restructuring.
Exhibit 2: Research Methodology
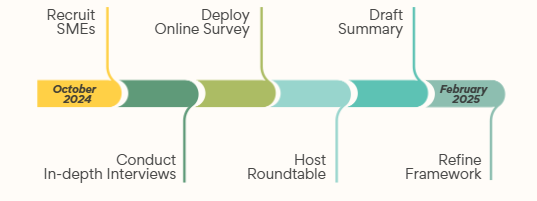
Over four months, we solicited individual and group SME perspectives on these public announcements and future professional implications. Virtual in-depth interviews queried opinions and perspectives on organizational changes and new expectations. Next, we deployed a brief internet survey to capture and prioritize internal stakeholders, key metrics, and functional impact expectations. Finally, Alkemi hosted a virtual SME roundtable discussion to brainstorm potential challenges and ways to convey functional impact.
Alkemi drafted a summary report which was circulated for review. Contributors shared input on findings, provided critical comments on implications, and refined the framework developed.
Eleven (11) pharmaceutical experts consented to join this research initiative (Exhibit 3).
Exhibit 3: Primary Research Sample
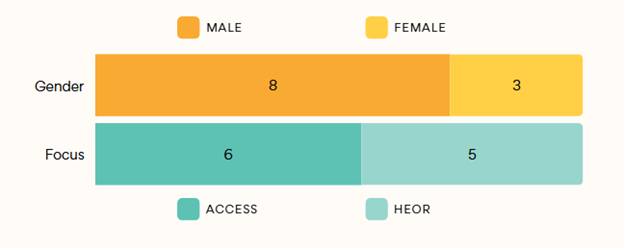
Participants from 9 organizations had a cumulative of 250 years of HEOR / Market Access experience.
Fortune 500 pharmaceutical companies account for 5 out of 9 organizations covered in research sample.
Five (5) key themes emerged from the interviews, internet survey, and virtual roundtable.
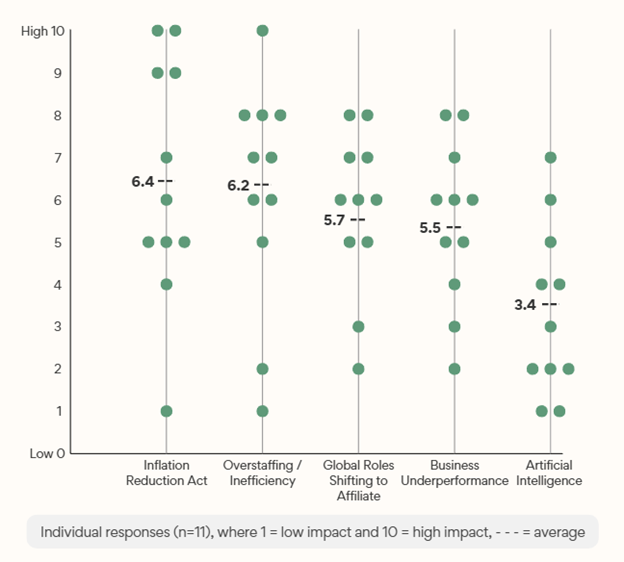
Participants were asked to comment on the rationale for the organizational changes and to rank 5 specific factors on a 10-point scale from least to most impact (Exhibit 4). Factors receiving the highest average impact scores represented external and internal threats to profitability: the Inflation Reduction Act and Overstaffing/ Inefficiency (as perceived by leadership).
Exhibit 4: Factors Impacting Recent Restructuring
Participants shared key changes they experienced due to restructurings (Exhibit 5). While restructuring and layoffs impacted other business functions, HEOR and Market Access associates were targeted, impacting structure, size, and functional reporting lines. This surprised our group of experts given the pharmaceutical imperative to demonstrate product value, and the fact that value strategies are the major focus of these functions.
Exhibit 5: Key Changes from Recent Restructuring
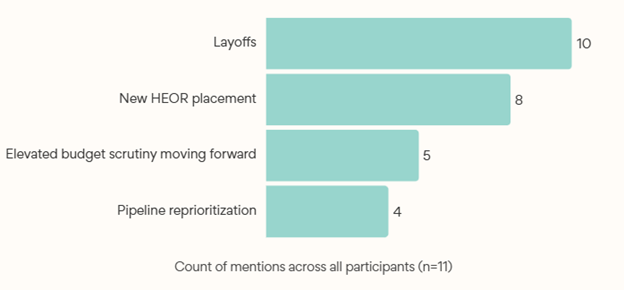
Participants reported that this restructuring feels different than past reorganizations due to reach and impact on HEOR.
Historically, pharmaceutical companies have adopted many governance structures for the HEOR function, including:
Prior to this restructuring, most experts in our focus group observed HEOR structured within Medical Affairs. Our research found the recent restructurings reversed this trend, shifting HEOR to sit within or alongside Market Access, aligned to a Commercial perspective.
Participants highlighted that increased HEOR alignment with Commercial functions coincides with a broader shift in external value pressures leading to new expectations of commercial impact. Management increasingly values HEOR activities by market outcomes achieved (e.g., access decisions, price achievement, and product adoption). Value leaders may be asked to justify budgets and resources for common HEOR activities in commercial terms:
While real world evidence (RWE) is an HEOR activity recognized as commercially impactful, some organizations have established separate RWE departments in Medical organizations.
Value leaders are likely to face increased scrutiny for resources across the product lifecycle. While a commercial impact metric, such as net price achievement, can be measured post-launch, meeting this goal requires a strong foundation of value planning and HEOR investment earlier in product development. Articulating a “return on investment” for pipeline HEOR resources is particularly difficult.
The internet survey and roundtable discussion identified a wide range of decision makers involved in the approval of HEOR budgets and resources, citing Commercial/Program Leads, Global Access, and Medical Affairs; however, this depended on organizational reporting, program stage, and financial processes. Further complicating this situation, HEOR experts within a Commercial organization may not have a direct path to influence early evidence resourcing determined by Clinical and Medical teams.
Leaders can use this framework to communicate HEOR impact regardless of product development stage.
Exhibit 6: Impact Communication Framework
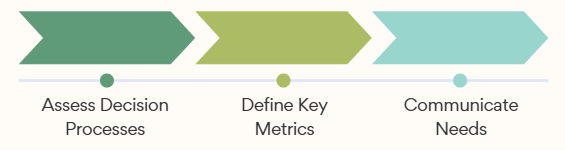
To impact commercial outcomes, Value leaders must implement this framework (Exhibit 6) across product development, beginning with creation of internal strategies to ensure the value proposition foundation is in place.
New leaders should first assess decision processes, which vary across organizations. This includes identifying key stakeholders who must recognize HEOR value: those controlling HEOR resources and collaborators who help advocate to decision makers. Organizational complexity has made this assessment more challenging. Ideally, Value leads work across functional groups throughout the product lifecycle to shape medical and commercial strategies.
The framework supports communication of appropriate metrics with decision makers.
Once stakeholders and decision-making processes applicable to HEOR value strategies are clear, the next step is to capture metrics to communicate both wins and progress toward commercial impact for the specific stakeholders previously identified at any given development stage. Consider a potential metric ‘trial design input’ for a pre-launch asset. Failure to communicate the impact of collecting patient outcomes or resource use data in trials is a missed opportunity for efficient evidence investment that could also thwart downstream commercial goals through low payer acceptance and negative health technology assessment outcomes.
Optimal channels to secure strategic buy-in and resources for value strategies will be specific to stakeholders and by organization. For those new in their Value function role or representing a new functional value group, meetings with cross-functional decision makers provide an opportunity to communicate how HEOR activities and collaborations across the product lifecycle enable the commercial value proposition. This internal engagement opens channels for effective communication with the correct stakeholders, using the correct impact metrics.
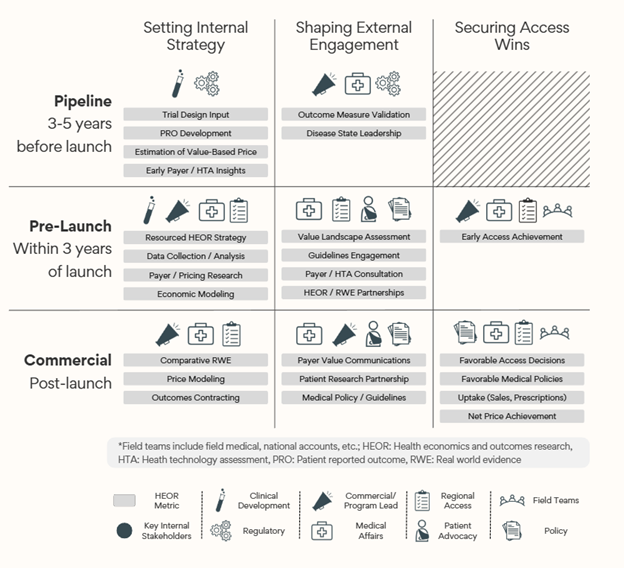
The value dashboard is an internal too to communicate commercial impact
Value functional leads are likely to face increased scrutiny as they seek to establish the value strategy early in product lifecycles.
To help communicate the rationale for HEOR investments throughout product development, we propose a Value Dashboard (Exhibit 7) which can be customized with key functional stakeholders, activities, and metrics which are aligned to organizational value strategies.
This tool is meant to be a visual representation of what implementing this impact communication framework may look like at your organization. For leaders with a narrower mandate, focusing on a particular vertical or horizontal of the dashboard may also be instructional.
Exhibit 7: Value Dashboard for Commercial Impact
To optimize the commercial impact of Value functions post-restructuring, we call on leader to:
Value functional leaders are experts in establishing value for external healthcare decision makers, however commercial impact, especially for Health Economics & Outcomes Research investments, also requires a “value story” for colleagues within their pharmaceutical organizations.
Use the framework and tool shared here to foster productive collaborations, demonstrate value, and advocate for resourcing.
Ask bold questions to embed value-aligned thinking across all functions of the organization and stages of product development.
Setting the Internal Strategy: “How can we build foundational evidence to establish the burden, unmet needs, and differentiated product value proposition?”
Shaping the External Market: “How can we engage thought leaders, patient advocates, payers, and policy makers to prepare the market for our launch value proposition?”
Securing Access Wins: “Are we continuing to differentiate and effectively communicate comparative value to achieve price and access targets?”


Schedule a consultation below. Together, we’ll look at your situation and explore your value proposition. If we can help, we’ll explain how. If not, we’ll connect you with a partner who can.